Periodontitis and Alzheimer's: A Bacterium Infects the Brain
Leggi in Italiano
In brief — Porphyromonas gingivalis from periodontitis reaches the brain haematogenously and via the trigeminal nerve, delivering its gingipains. In the brain it generates neuroinflammation, amyloid plaques, neurofibrillary tangles and synaptic loss — the hallmarks of Alzheimer’s — even in young, healthy animals.
The brain has always been considered a biological “sanctuary.” It is difficult to imagine it vulnerable to what comes from below, with the exception of the circulatory problems known to all.
In 2019, a large group of researchers published in Science Advances a highly significant study on Alzheimer’s disease (DOI: 10.1126/sciadv.aau3333).
The brains of patients who had died with Alzheimer’s contained Porphyromonas gingivalis — the key bacterium of periodontal disease — and its gingipains, the proteolytic enzymes this bacterium uses to destroy periodontal tissues and suppress the immune response. And the levels of gingipains in the brain correlated with those of Alzheimer’s disease markers: tau pathology and ubiquitin.
For many years now, the link between Alzheimer’s and periodontitis has been recognised. One proposed mechanism is neuroinflammation caused by increased inflammatory mediators (cytokines, metalloproteinases, membrane lipopolysaccharides) capable of crossing the blood-brain barrier — which normally protects the central nervous system from toxic blood contents.
The direct infection mechanism described in this paper is more concerning. The fact that a dental bacterium can reach the brain and colonise it, with all attendant consequences, should make us reflect once more on the central role of dental prevention in general health.
How much chronic disease — weighing heavily on public health costs — could be prevented with even simple dental interventions, such as routine periodontal hygiene?
Certainly not all, since these are always multifactorial diseases. Yet a significant portion of pathology could be avoided with this simple habit.
Porphyromonas Gingivalis: From the Periodontal Pocket to the Brain
Every periodontist knows P. gingivalis. It is the pathogen that orchestrates the destruction of the deep periodontium. It produces gingipains — proteases (enzymes) that digest human proteins, subvert the immune response and cause chronic inflammation. We have known this for decades. P. gingivalis is an integral part of Sigmund Socransky’s famous red complex. (See also: Integrin Beta-1: Key to Porphyromonas Invasion)
What we have known for less time is that this bacterium does not remain confined to the mouth, but has an aggressive capacity, when it develops unchecked, that extends to the entire body.
In patients with periodontal disease, during mastication, brushing — any manipulation of inflamed tissues — P. gingivalis enters the bloodstream. This is the transient bacteraemia that every periodontist knows and combats with antibiotic prophylaxis in at-risk patients, such as those with cardiac conditions.
But the destination of that bacterium, once in the blood, is less well known.
How it reaches the brain
Researchers have identified at least two routes.
The first is the blood-brain barrier. Under normal conditions, this barrier is almost impermeable. But bacteraemia from P. gingivalis increases its permeability. A study published in International Journal of Oral Science demonstrated that gingipains increase the passage of molecules through endothelial cells, opening the door to the bacterium. (https://doi.org/10.1038/s41368-022-00215-y)
The second route is even more concerning, if confirmed.
The trigeminal nerve — famous for its namesake neuralgia — is the fifth cranial nerve and directly innervates periodontal tissues.
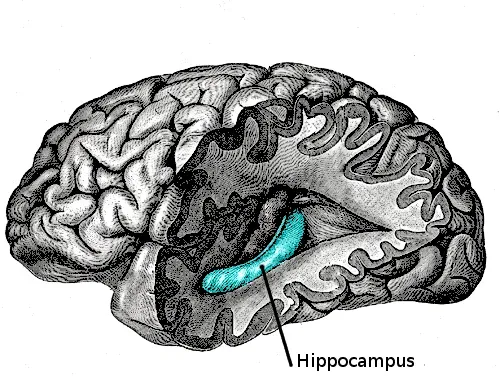
A study in the Journal of Advanced Research demonstrated in rats that extracellular vesicles produced by P. gingivalis — small cellular “bubbles” carrying gingipains — travel along nerve fibres, reach the trigeminal ganglion and from there the hippocampus (at the centre of the brain). Neurectomy (cutting) of the trigeminal nerve blocks this pathway. (https://doi.org/10.1016/j.jare.2023.02.006)
Experimental Evidence in Animal Models
Murine (mouse) models are widely tested and validated. They provide solid evidence.
In 2018, Ilievski and colleagues (https://doi.org/10.1371/journal.pone.0204941) applied P. gingivalis orally to mice for twenty-two weeks — simulating a chronic periodontal infection. The experimental periodontitis produced the following effects in the mice’s hippocampi:
- Neuroinflammation with increased TNFα, IL-1β and IL-6
- Neurodegeneration with reduced intact neurons
- Extracellular beta-amyloid 42 production
- Tau protein phosphorylation
- Neurofibrillary tangles
These are all pathognomonic features of Alzheimer’s — but in young, genetically normal mice, produced solely by oral P. gingivalis infection.
In 2022, Hao and colleagues (https://doi.org/10.3389/fimmu.2022.816640) added another piece to the mosaic. Their experimental data suggest that oral P. gingivalis infection accelerates cognitive deterioration and beta-amyloid accumulation, and that the mechanism operates through hyperactivation of complement C1q, which amplifies microglial activation, cerebral inflammation and — crucially — causes synaptic phagocytosis. The periodontitis bacterium, in essence, induces the brain to self-eliminate its own neural connections.
A 2024 Australian study by Ciccotosto and colleagues in the Journal of Infectious Diseases reports that chronic oral inoculation of P. gingivalis in mice causes a series of cerebral pathologies: neuronal damage, astrocyte and microglial activation, high inflammatory cytokine levels, amyloid plaques and hyperphosphorylated tau. (https://doi.org/10.1093/infdis/jiae286)
Evidence in Humans: Meta-analyses
The epidemiological data in humans confirm the association between P. gingivalis and Alzheimer’s.
The meta-analysis by Asher and colleagues, published in the Journal of the American Geriatrics Society in 2022, analysed forty-seven longitudinal studies. Periodontitis and tooth loss in humans are statistically associated with both cognitive decline and dementia (https://doi.org/10.1111/jgs.17978).
The meta-analysis by Dibello and colleagues, published in GeroScience in 2024, extended the analysis to forty-six studies. Periodontitis is associated with an increased risk of cognitive deterioration and dementia (https://doi.org/10.1007/s11357-024-01243-8).
The numbers have the known limitations of observational research, but they all point in the same direction. And the experimentally demonstrated biological mechanisms are solid.
Beyond Alzheimer’s: Parkinson’s
In 2024, Ermini, Dominy and colleagues — the same group behind the 2019 Science Advances study — published in NPJ Parkinson’s Disease a study in which gingipains were identified in the substantia nigra of brains from Parkinson’s disease patients. (https://doi.org/10.1038/s41531-024-00705-2) Gingipains were abundant in dopaminergic neurons, localised in the perinuclear cytoplasm, neuromelanin, mitochondria and nucleus. Gingipains interact with Parkinson’s-associated proteins.
The periodontitis bacterium, therefore, appears to be not only a risk factor for Alzheimer’s, but for Parkinson’s as well.
What This Means for Patients
For those who work in the mouth every day, these data carry concrete significance.
Periodontitis is not a strictly dental disease. It is a chronic inflammatory disease with systemic consequences. The patient often comes to the practice with bleeding gums and thinks it is nothing — that patient has a chronic infection from which bacteria enter the bloodstream. Some of those bacteria potentially reach the brain. And there they produce the same enzymes that in the mouth destroy bone.
Treating periodontitis is not just a therapy aimed at saving teeth. It is reduction of systemic bacterial burden. It is reduction of chronic inflammation. It is protection of the brain.
We cannot tell patients that treating periodontitis prevents Alzheimer’s. The direct proof of a protective effect in humans does not yet exist, although we have statistical data supporting this hypothesis. But we can tell them the truth: the bacterium that causes periodontitis has been found in the brains of Alzheimer’s patients. The levels of its enzymes correlate with the levels of disease markers. And in animal models, periodontal infection produces the same brain changes as Alzheimer’s.
See also
- HPV in the Oral Cavity: What You Need to Know — The role of P. gingivalis gingipains in oropharyngeal cancer
- Periodontitis and Pregnancy: A Link Most People Ignore — Systemic implications of periodontitis in pregnancy
- Do Oral Bacteria Control Your Weight? — How oral bacterial metabolites reach the brain
- 30 Years of the Relationship Between Teeth and General Health
FAQ
- What is the link between periodontitis and Alzheimer's?
- Porphyromonas gingivalis, the principal bacterium of chronic periodontitis, has been identified in the brains of Alzheimer's patients. Its proteolytic enzymes, gingipains, correlate with disease markers. In animal models, chronic oral infection produces neuroinflammation, amyloid plaques and neurofibrillary tangles — the hallmarks of Alzheimer's.
- How does a mouth bacterium reach the brain?
- Through at least two routes: the blood-brain barrier, whose permeability increases during periodontal bacteraemia due to gingipains acting on endothelial cells, and the trigeminal nerve, which directly innervates periodontal tissues and transports the bacterium's extracellular vesicles to the hippocampus.
- Does tooth loss increase the risk of dementia?
- Meta-analyses of longitudinal studies indicate that tooth loss is an independent risk factor for both cognitive decline and dementia. Partial tooth loss weighs more on cognitive decline; total tooth loss weighs more on dementia.
- Can treating periodontitis prevent Alzheimer's?
- Direct proof that periodontal treatment prevents Alzheimer's in humans does not yet exist. However, the biological mechanisms are clear: periodontitis maintains a chronic infection that fuels bacteraemia, systemic inflammation and bacterial translocation to the brain. Reducing bacterial burden and inflammation is biologically consistent with risk reduction.
- Is the periodontitis bacterium also involved in Parkinson's?
- Yes. P. gingivalis gingipains have been identified in dopaminergic neurons of the substantia nigra in Parkinson's brains. In vitro, lysine-gingipain cleaves alpha-synuclein, generating fragments implicated in the pathological aggregation characteristic of the disease.
References
Looking for a specialist?
Parodontologia a Frosinone →Diagnosi e trattamento della parodontite nello Studio Denti Più
Need a professional opinion?
Book an appointment at Dr. Bruschi's practice in Frosinone. First visit includes full diagnosis and personalised treatment plan.
Stay Updated
New articles on periodontology, implantology and oral surgery — delivered to your inbox.
Comments
Loading comments...
Leave a comment