Peri-implantitis and Periodontitis: Microbiota Compared
Leggi in Italiano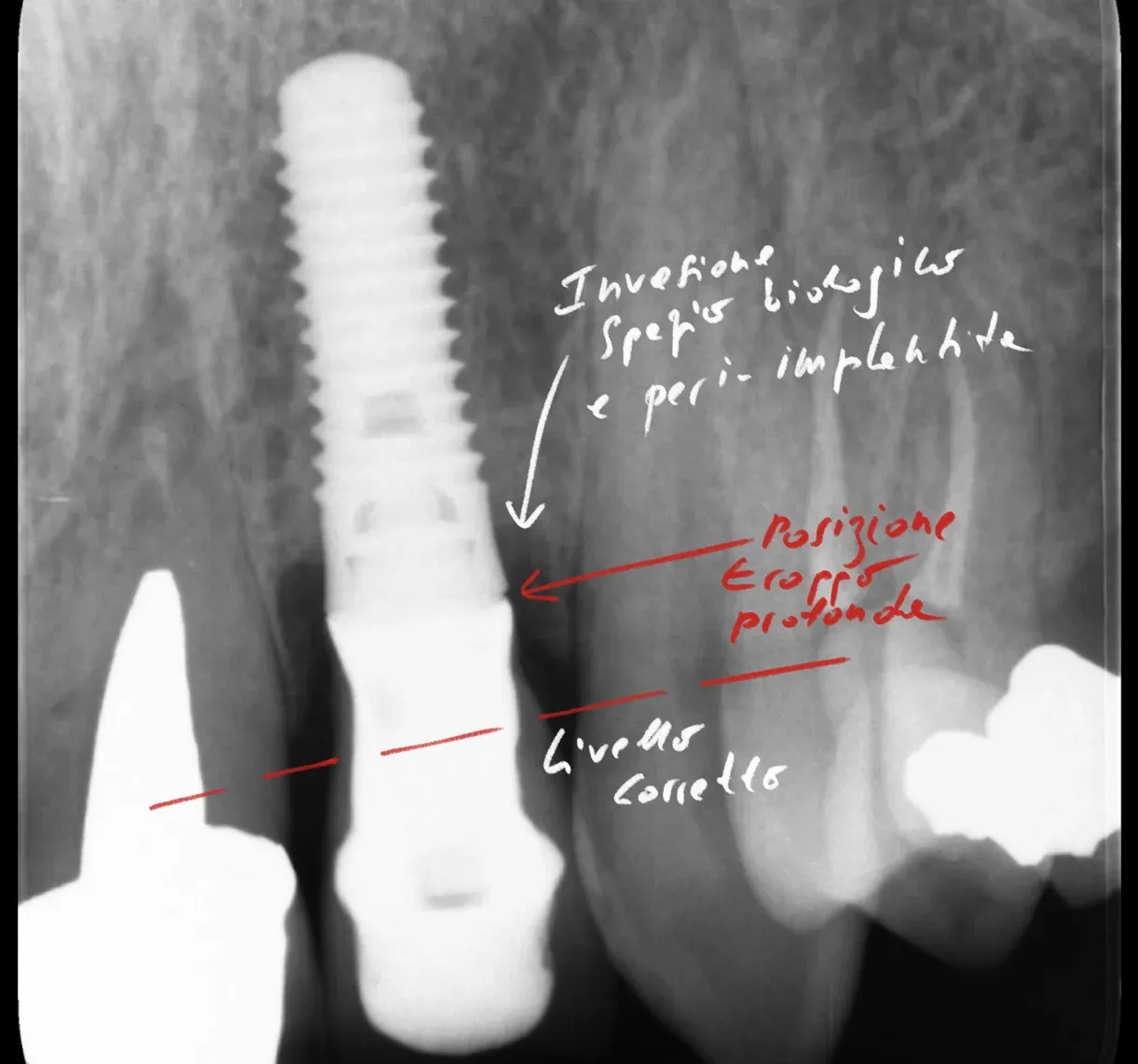
In brief — Peri-implantitis and periodontitis share many bacteria but diverge in relative composition and virulence. The implant surface (titanium) selects distinct microbial communities from natural teeth. Pathogenic mechanisms differ: motility and tissue invasion play more central roles in peri-implantitis.
Similarities, differences and pathogenic mechanisms
Shared microbial groups
Peri-implantitis and periodontitis harbour microbial communities that draw from the same oral microbiota repertoire: both diseases are polymicrobial, and many of the classic aetiological agents are found in both niches.
Furthermore, in both pathologies many factors contribute to establishing a pathological condition. We speak, therefore, of multifactoriality.
Yet what varies — and what matters — are the relative abundances of individual species and their level of activity.
Species of the genera Porphyromonas, Tannerella and Treponema are consistently identified in both peri-implantitis and periodontal disease sites [1]. Similarly, Fusobacterium and Streptococcus emerge as predominant genera in both conditions in direct comparisons [2]. Even more interesting is that virulence-related gene expression profiles are substantially overlapping at the mRNA level, even when taxonomic profiles diverge [3].
For a closer look at how Porphyromonas gingivalis interacts with host cells, see Integrin Beta-1: Key to Porphyromonas Invasion.
Distinct taxa and predominance
No species is universally exclusive to peri-implantitis.
This datum in itself tells us something important about the ecology of these diseases: there is no single culprit, no sentinel pathogen (as A. actinomycetemcomitans might be in juvenile forms of periodontitis) that marks a clear boundary between the two conditions. Yet some species recur as discriminants with sufficient frequency to build a distinct microbial identity.
On the role of A. actinomycetemcomitans and the classification of aggressive forms, I wrote here: The Migrant Bacterium — AA for African Aggressive and here: 2017 Classification of Periodontal Diseases: Where Did Juvenile Go?.
Species enriched or active in peri-implantitis
Analyses identify Solobacterium moorei and Prevotella denticola as two actors with high transcriptional activity, classified as keystone bacteria of peri-implantitis [4]. Alongside them, Prevotella nigrescens and other representatives of the genus Prevotella — together with more recently identified species such as Olsenella uli, Treponema parvum and Slackia exigua — are significantly more abundant in peri-implantitis patient groups [5][6]. Metagenomics has added a further layer of complexity, implicating specific subspecies of Fusobacterium nucleatum as key colonisers in peri-implant disease progression [7].
Species relatively associated with periodontitis
The intra-individual comparison returns a mirror image: some members of Peptostreptococcaceae and Desulfomicrobium orale appear in greater proportions in periodontitis sites compared to peri-implantitis sites [5]. A relevant asymmetry, suggesting that the surface on which biofilm grows — titanium or root cementum — is not a neutral detail, but a factor that steers bacterial ecological selection.
No pathognomonic species
Systematic reviews agree on one point: no microorganism is specifically and consistently associated with peri-implantitis. Some species — Aggregatibacter actinomycetemcomitans, Prevotella intermedia — show elevated prevalence in diseased implant sites compared to healthy implants, but variability across studies remains high [1].
Diversity and composition
Data on microbial diversity are, in any case, contradictory across studies. Some report greater taxonomic richness in peri-implantitis; others find no significant differences; systematic reviews, which by nature rarely conclude sharply, lean towards heterogeneity.
Pathogenic mechanisms and biofilm formation
This is where the story becomes more interesting. It is not just about which species inhabit the peri-implant mucosa, but about how those microorganisms behave, and how they interact with each other.
Studies converge on one conclusion: peri-implantitis is not simply periodontitis implanted on titanium.
Comparisons have identified bacteria with high specific activity in peri-implantitis and greater expression of certain genes — including the plasmin receptor and glyceraldehyde-3-phosphate dehydrogenase — that may influence interactions with host tissues and modulate clinical disease manifestations [4].
In peri-implantitis, functions related to flagellar assembly and epithelial interaction are enriched, suggesting that microbial motility — and therefore tissue invasion capacity — plays a peculiar role in implant site disease [8]. The topic of cellular invasion by gingival bacteria is discussed in detail in this article.
The surface differences between implant and tooth — material properties, peri-implant sulcus anatomy — appear to shape colonisation and subsequent biofilm ecology from the earliest stages [5][2]. No surprise: a surface’s topography and chemistry determine who colonises it first, and the first coloniser steers what follows. On how blood interacts with the titanium surface in the first minutes, read “Bind me!” Blood and Titanium: Love Is Born in Five Minutes.
The overall picture emerging from the literature is one of two diseases that draw from the same oral repertoire but diverge in who becomes dominant, how taxa interact within the biofilm, and which microbial functions prevail at the implant site.
Systemic host factors — immunity, metabolism, medications — also contribute to steering the outcome. I discuss this in Failure Is Rare: Watch Out for Immunity, Metabolism and Medications.
Frequently asked questions
Are the bacteria the same in peri-implantitis and periodontitis? Yes and no. They share many species — Porphyromonas, Fusobacterium, Streptococcus — but relative abundances differ. Peri-implantitis enriches Solobacterium moorei and Prevotella denticola; periodontitis favours certain Peptostreptococcaceae. It is not the repertoire that changes, but the hierarchy.
Is there a specific bacterium for peri-implantitis? No. No microorganism is pathognomonic for peri-implantitis. This is the most relevant absence in the literature: the disease is plural, not monocausal. Even titanium does not select a single dominant species, but steers an entire ecosystem.
Why does titanium select different communities from natural teeth? Surface topography and chemistry determine initial colonisation. Titanium is chemically inert, lacking protein ligands like root cementum. This steers from the outset which bacterium arrives first, and the first coloniser writes the rules of the ecology that follows.
Is bacterial motility really more relevant in peri-implantitis? Yes. Metagenomic studies show enrichment of flagellar assembly genes at peri-implantitis sites. This suggests that in implant disease, the ability to move and invade tissues plays a more central role than in classic periodontitis.
Can an individual have both peri-implantitis and periodontitis at the same time? Biologically yes, even at the same oral site. Intra-individual studies have found microbial differences between teeth and implants in the same patient, suggesting that each surface creates its own microbiota, even when the host is identical.
If genetic virulence is overlapping, what explains the clinical differences? Good question. The relative gene expression profile (mRNA) is overlapping, but absolute abundances and community composition differ. Moreover, host factors — immunity, metabolism, medications — steer the clinical course. Peri-implantitis is not a “more aggressive” version; it is a different ecological configuration.
References
-
Komatsu K et al. Discriminating Microbial Community Structure Between Peri-Implantitis and Periodontitis With Integrated Metagenomic, Metatranscriptomic, and Network Analysis. Front Cell Infect Microbiol 2020. doi:10.3389/fcimb.2020.596490
-
Maruyama N et al. Intraindividual variation in core microbiota in peri-implantitis and periodontitis. Sci Rep 2015. doi:10.1038/srep06602
-
Casarin et al. Unraveling the Microbial Mosaic and Inflammatory Core of Peri-implantitis. J Dent Res 2026. doi:10.1177/00220345251344585
-
Yu et al. Microbiome of periodontitis and peri-implantitis before and after therapy: Long-read 16S rRNA gene amplicon sequencing. J Periodontal Res 2024. doi:10.1111/jre.13269
-
Yu X-L et al. Intra-oral single-site comparisons of periodontal and peri-implant microbiota in health and disease. Clin Oral Implants Res 2019. doi:10.1111/clr.13459
-
Sahrmann P et al. The Microbiome of Peri-Implantitis: A Systematic Review and Meta-Analysis. Microorganisms 2020. doi:10.3390/microorganisms8050661
-
Parga A et al. Do Concurrent Peri-Implantitis and Periodontitis Share Their Microbiotas? A Pilot Study. Dent J 2024. doi:10.3390/dj12040113
-
Song et al. Metagenomic analysis of healthy and diseased peri-implant microbiome under different periodontal conditions: a cross-sectional study. BMC Oral Health 2024. doi:10.1186/s12903-023-03442-9
FAQ
- Are the bacteria the same in peri-implantitis and periodontitis?
- Yes and no. They share many species — Porphyromonas, Fusobacterium, Streptococcus — but relative abundances differ. Peri-implantitis enriches Solobacterium moorei and Prevotella denticola; periodontitis favours certain Peptostreptococcaceae. It is not the repertoire that changes, but the hierarchy.
- Is there a specific bacterium for peri-implantitis?
- No. No microorganism is pathognomonic for peri-implantitis. This is the most relevant absence in the literature: the disease is plural, not monocausal. Even titanium does not select a single dominant species, but steers an entire ecosystem.
- Why does titanium select different communities from natural teeth?
- Surface topography and chemistry determine initial colonisation. Titanium is chemically inert, lacking protein ligands like root cementum. This steers from the outset which bacterium arrives first, and the first coloniser writes the rules of the ecology that follows.
- Is bacterial motility really more relevant in peri-implantitis?
- Yes. Metagenomic studies show enrichment of flagellar assembly genes at peri-implantitis sites. This suggests that in implant disease, the ability to move and invade tissues plays a more central role than in classic periodontitis.
- Can an individual have both peri-implantitis and periodontitis at the same time?
- Biologically yes, even at the same oral site. Intra-individual studies have found microbial differences between teeth and implants in the same patient, suggesting that each surface creates its own microbiota, even when the host is identical.
- If genetic virulence is overlapping, what explains the clinical differences?
- Good question. The relative gene expression profile (mRNA) is overlapping, but absolute abundances and community composition differ. Moreover, host factors — immunity, metabolism, medications — steer the clinical course. Peri-implantitis is not a "more aggressive" version; it is a different ecological configuration.
Looking for a specialist?
Parodontologia a Frosinone →Diagnosi e trattamento della parodontite nello Studio Denti Più
Need a professional opinion?
Book an appointment at Dr. Bruschi's practice in Frosinone. First visit includes full diagnosis and personalised treatment plan.
Stai valutando un impianto dentale?
Ho scritto una guida in 8 capitoli che spiega tutto quello che un paziente dovrebbe sapere prima di sedersi in poltrona. Niente marketing — solo fatti, casi studio e una checklist per fare le domande giuste.
Scarica la guidaStay Updated
New articles on periodontology, implantology and oral surgery — delivered to your inbox.
Comments
Loading comments...
Leave a comment