Treponema denticola: The Spirochete of the Red Complex
Leggi in Italiano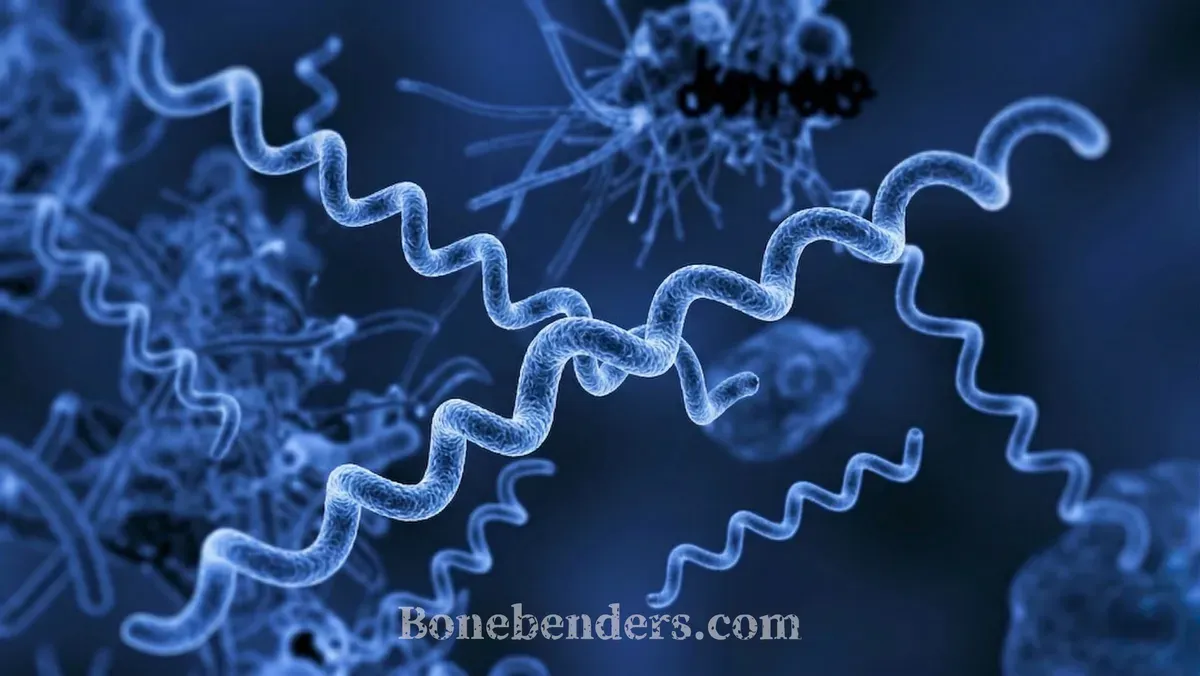
In brief — Treponema denticola is an anaerobic oral spirochete belonging to Socransky’s red complex. It stands apart from other periodontal pathogens by its motility: periplasmic flagella that allow it to penetrate tissues where immotile bacteria cannot reach. It carries a surface protease, dentilisin, which degrades host defences and facilitates coaggregation with the other red complex members. Emerging evidence links it to neurodegenerative diseases.
In Socransky’s red complex, each member has a role. Porphyromonas gingivalis is the director: it orchestrates dysbiosis, subverts defences, reshapes the environment. Tannerella forsythia is the silent accomplice, present wherever the damage has already been done.
Treponema denticola is something else entirely. It is the one that moves. Picture a micro-serpent screwing its way through the biofilm of the periodontal pocket.
An ancient form
Spirochetes are among the oldest bacteria on the planet. Their helical shape is no accident: it is an engineering solution. While most bacteria swim in free fluids using external flagella, spirochetes solved a different problem — moving through viscous, dense, semi-solid environments.
T. denticola has a three-layered structure. At the centre lies the protoplasmic cylinder, containing the genetic material and metabolic machinery. Around it, in the periplasmic space, run the flagella — called periplasmic flagella or endoflagella. Outside, an outer sheath wraps everything like an elastic membrane.
The periplasmic flagella are the heart of the system. Anchored at the cell poles and extending toward the centre, overlapping. When they rotate, they generate a wave that propagates along the entire bacterial body. The result is a corkscrew movement that allows the spirochete to cross mucus, crevicular fluid, gingival connective tissue. Kurniyati and colleagues demonstrated in 2022 that sigma factor FliA (TDE2683) regulates late flagellar gene expression and controls flagellar symmetry: when altered, motility collapses.
This movement is not random. T. denticola is chemotactic: it moves toward nutrients. Ruby and colleagues showed that even simple glucose induces a motility-dependent chemotactic response. The bacterium does not wander aimlessly: it seeks, finds, penetrates.
The outer sheath and its weapons
The outer sheath of T. denticola is not a simple envelope. It is an armed platform.
Two proteins dominate its surface.
Msp (major sheath protein) is a porin — a protein that forms channels in the membrane. But Msp does far more: it is cytotoxic to gingival fibroblasts, alters epithelial cell permeability, and contributes to bacterial adhesion to host tissues. Sela, in his 2001 review in Critical Reviews in Oral Biology and Medicine, called it one of the principal virulence determinants.
Dentilisin is a surface protease with chymotrypsin-like activity. It degrades extracellular matrix proteins — fibronectin, laminin, complement components — and disrupts tight junctions between epithelial cells, paving the way for bacterial invasion. But its role does not stop there.
Kokubu and colleagues demonstrated in 2021 that dentilisin also facilitates crawling — creeping movement on solid surfaces. Mutant strains lacking dentilisin showed smaller colonies and reduced displacement speed. The protease, therefore, is not just a weapon: it is also a locomotion engine.
The alliance with Porphyromonas gingivalis
The red complex forms a metabolic consortium.
Zhu and colleagues, in 2013 in PLoS ONE, documented a strong synergy between P. gingivalis and T. denticola in polymicrobial biofilm formation. In co-culture, both species significantly increased biofilm biovolume, mean thickness and maximum thickness. P. gingivalis gingipains proved essential for this synergy: a gingipain-deficient mutant was unable to form polymicrobial biofilms with T. denticola.
In 2020, Kin and colleagues revealed the underlying metabolic mechanism, publishing in Journal of Oral Microbiology. P. gingivalis hydrolyses glycine-containing peptides and produces free glycine, which T. denticola uses as a primary energy and carbon source. In return, P. gingivalis receives succinate. This is genuine metabolic cooperation: the two bacteria share resources, and together become more efficient than either would be alone.
Richard Darveau, in 2010 in Nature Reviews Microbiology, described periodontitis as a polymicrobial disruption of host homeostasis. T. denticola is a central player in this phenomenon. It is a powerful amplifier of the pathogenicity of the other bacteria involved.
Dentilisin also plays a role in coaggregation with Tannerella forsythia. Sano and colleagues showed in 2014 in Anaerobe that T. denticola strains lacking dentilisin lost the ability to coaggregate with T. forsythia. Not through direct proteolytic effect, but probably through maturation of surface ligands. An indirect, elegant mechanism.
Beyond the mouth
Like P. gingivalis, T. denticola is also suspected of reaching sites distant from the oral cavity.
Pisani and colleagues, in 2023 in International Journal of Environmental Research and Public Health, proposed a suggestive hypothesis: T. denticola might cause neurodegeneration in the midbrain via the periodontal route. The spirochete is neurotropic — it can travel along axons of periodontal ligament peripheral nerves, reach the locus coeruleus and from there extend damage to the cortex. The proposed mechanisms include cytoskeletal damage, axonal transport disruption and mitochondrial dysfunction.
After all, its cousin Treponema pallidum, the causative agent of syphilis, has well-established neurotropism and causes neurosyphilis.
One more piece of data: Mahendrarajan and colleagues in 2025, in Frontiers in Oral Health, described the role of outer membrane vesicles (OMVs) from red complex bacteria — T. denticola included — as long-range virulence vectors, capable of influencing atherosclerosis, rheumatoid arthritis and diabetes mellitus.
These are hypotheses, not certainties. But the direction of research is clear: what happens in the gingival sulcus does not stay in the gingival sulcus.
What it means for clinicians
T. denticola is an obligate anaerobe. It lives only where oxygen cannot reach — in the depths of periodontal pockets, in mature subgingival biofilm, in spaces where tissue is already compromised. This fact alone dictates the therapeutic strategy.
Mechanical biofilm removal — scaling, root planing, resective or regenerative surgery when indicated — is the most direct way to reduce its burden. T. denticola does not survive in a clean, shallow sulcus. Regular periodontal maintenance prevents recolonisation.
Dashper and colleagues, in their 2010 review in Journal of Dental Research, emphasised that T. denticola also possesses non-classical virulence determinants — the ability to interact with other pathogens and with the host in ways that promote disease progression. This explains why, in clinical practice, reducing T. denticola is often accompanied by clinical improvement that exceeds the numerical weight of the single bacterium.
Holt and Ebersole had anticipated this in 2005 in Periodontology 2000: the red complex is a consortium, and must be treated as such. You do not eliminate one member at a time. You destroy the habitat.
Parallels between T. pallidum (syphilis) and T. denticola
Both belong to phylum Spirochaetota, family Treponemataceae, genus Treponema. The phylogenetic proximity implies shared cellular architecture, analogous motility mechanisms, and convergent pathogenic strategies.
The morphology is identical in form: helical body, 6–20 µm, with periplasmic flagella conferring the characteristic corkscrew motility. This is active motility even in viscous environments — connective tissue, extracellular matrix — that distinguishes spirochetes from most periodontal bacteria.
Immune evasion
T. pallidum is the master of evasion: an outer surface nearly devoid of exposed transmembrane proteins, low immunogenicity, the ability to persist for decades. T. denticola uses similar strategies — it degrades IgG and IgA immunoglobulins through specific proteases, inhibits neutrophil chemotaxis, and survives intracellularly in fibroblasts.
Protease production
Dentilisin of T. denticola — a cysteine protease — has direct cytotoxic activity, degrades extracellular matrix components and activates host MMPs. T. pallidum also expresses proteases that facilitate tissue dissemination.
References
- Dashper SG, Seers CA, Tan KH, Reynolds EC. Virulence factors of the oral spirochete Treponema denticola. J Dent Res. 2011;90(6):691-703. DOI
- Ishihara K. Virulence factors of Treponema denticola. Periodontol 2000. 2010;54(1):117-35. DOI
- Sela MN. Role of Treponema denticola in periodontal diseases. Crit Rev Oral Biol Med. 2001;12(5):399-413. DOI
- Holt SC, Ebersole JL. Porphyromonas gingivalis, Treponema denticola, and Tannerella forsythia: the “red complex”. Periodontol 2000. 2005;38:72-122. DOI
- Darveau RP. Periodontitis: a polymicrobial disruption of host homeostasis. Nat Rev Microbiol. 2010;8(7):481-90. DOI
- Zhu Y, Dashper SG, Chen YY, Crawford S, Slakeski N, Reynolds EC. Porphyromonas gingivalis and Treponema denticola synergistic polymicrobial biofilm development. PLoS One. 2013;8(8):e71727. DOI
- Kin LX, Butler CA, Slakeski N, Hoffmann B, Dashper SG, Reynolds EC. Metabolic cooperativity between Porphyromonas gingivalis and Treponema denticola. J Oral Microbiol. 2020;12(1):1808750. DOI
- Kurniyati K, Chang Y, Liu J, Li C. Identification and characterization of the alternative σ28 factor in Treponema denticola. J Bacteriol. 2022;204(9):e0024822. DOI
- Kokubu E, Kikuchi Y, Okamoto-Shibayama K, Nakamura S, Ishihara K. Crawling motility of Treponema denticola modulated by outer sheath protein. Microbiol Immunol. 2021;65(12):551-558. DOI
- Sano Y, Okamoto-Shibayama K, Tanaka K, Ito R, Shintani S, Yakushiji M, Ishihara K. Dentilisin involvement in coaggregation between Treponema denticola and Tannerella forsythia. Anaerobe. 2014;30:45-50. DOI
- Pisani F, Pisani V, Arcangeli F, Harding A, Singhrao SK. Treponema denticola has the potential to cause neurodegeneration in the midbrain via the periodontal route of infection—Narrative review. Int J Environ Res Public Health. 2023;20(11):6049. DOI
- Mahendrarajan V, Lazarus HPS, Muthukaliannan GK, Varghese S, Easwaran N. Membrane vesicles from Red Complex bacteria: key players in oral pathogenesis, immune disruption, systemic diseases, and therapeutic insights. Front Oral Health. 2025;6:1607931. DOI
FAQ
- What is Treponema denticola?
- It is a Gram-negative, obligate anaerobic spirochete and a member of Socransky's red complex. It inhabits the deep gingival sulcus and is strongly associated with chronic periodontitis.
- Why is Treponema denticola dangerous for gums?
- It possesses periplasmic flagella that allow tissue penetration, a protease called dentilisin that degrades host proteins, and the ability to evade the immune system. It also cooperates synergistically with Porphyromonas gingivalis.
- Can Treponema denticola cause diseases outside the mouth?
- Recent studies associate it with neurodegeneration (Alzheimer's), atherosclerosis and pregnancy complications. Its ability to invade tissues and travel along peripheral nerves makes it a plausible suspect.
- How can Treponema denticola be controlled?
- Through thorough oral hygiene, professional periodontal therapy and regular maintenance. As an obligate anaerobe, mechanical removal of subgingival biofilm is the most effective method to reduce its burden.
References
- https://doi.org/10.1177/0022034510385242
- https://doi.org/10.1111/j.1600-0757.2009.00345.x
- https://doi.org/10.1177/10454411010120050301
- https://doi.org/10.1111/j.1600-0757.2005.00113.x
- https://doi.org/10.1038/nrmicro2337
- https://doi.org/10.1371/journal.pone.0071727
- https://doi.org/10.1080/20002297.2020.1808750
- https://doi.org/10.1128/jb.00248-22
- https://doi.org/10.1111/1348-0421.12940
Looking for a specialist?
Parodontologia a Frosinone →Diagnosi e trattamento della parodontite nello Studio Denti Più
Need a professional opinion?
Book an appointment at Dr. Bruschi's practice in Frosinone. First visit includes full diagnosis and personalised treatment plan.
Stay Updated
New articles on periodontology, implantology and oral surgery — delivered to your inbox.
Comments
Loading comments...
Leave a comment