Streptococcus dentisani: The Good Bacterium That Protects Your Teeth
Leggi in Italiano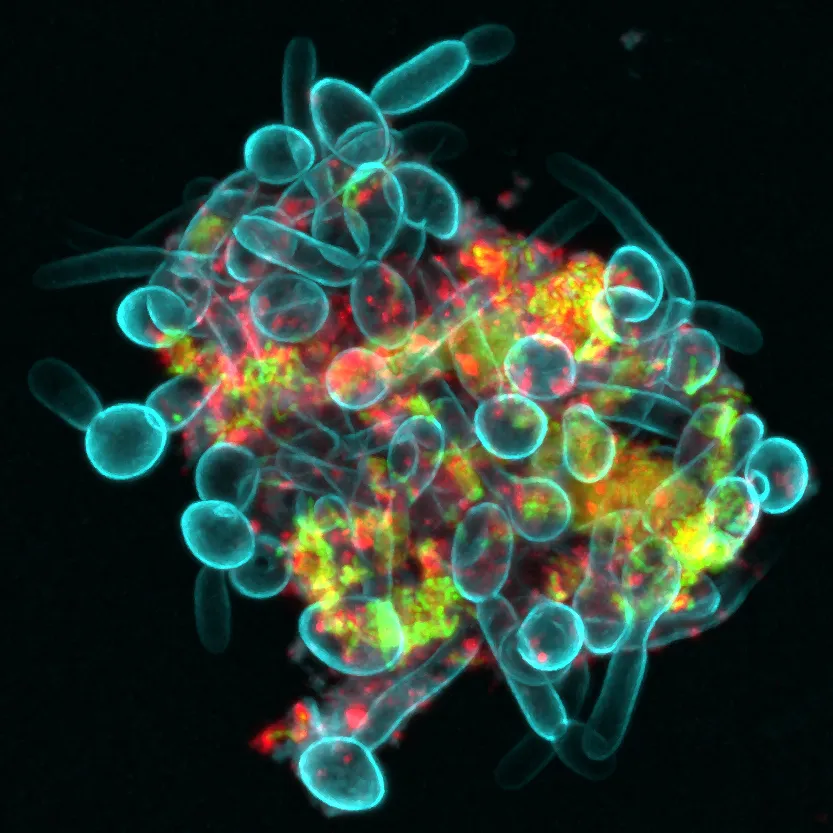
Around seven hundred bacterial species inhabit the human mouth. Most of them do no harm. Some are pathobionts: they turn aggressive when certain conditions arise. The classic example is the periodontal pocket.
Some do a great deal of good. One, in particular, seems to do precisely what is needed: kill pathogenic bacteria and neutralise the acid that destroys enamel. Its name is Streptococcus dentisani.
The discovery
In 2013, Alex Mira’s group at the FISABIO Foundation in Valencia described a new streptococcal species isolated from the tooth surfaces of caries-free individuals. Two strains — 7746 and 7747 — showed a genomic profile distinct from all known species of the mitis group, with ANI (Average Nucleotide Identity) values below 91% compared with S. oralis and S. mitis [1]. The species was named Streptococcus dentisani.
Three years later, Jensen and colleagues reclassified the bacterium as Streptococcus oralis subsp. dentisani on the basis of broader phylogenomic analyses [2]. The original name, however, has persisted in common usage and in the probiotic literature.
Two weapons, one bacterium
What makes S. dentisani interesting is not just where it lives, but what it does. López-López et al. (2017) described a dual mechanism of action [3].
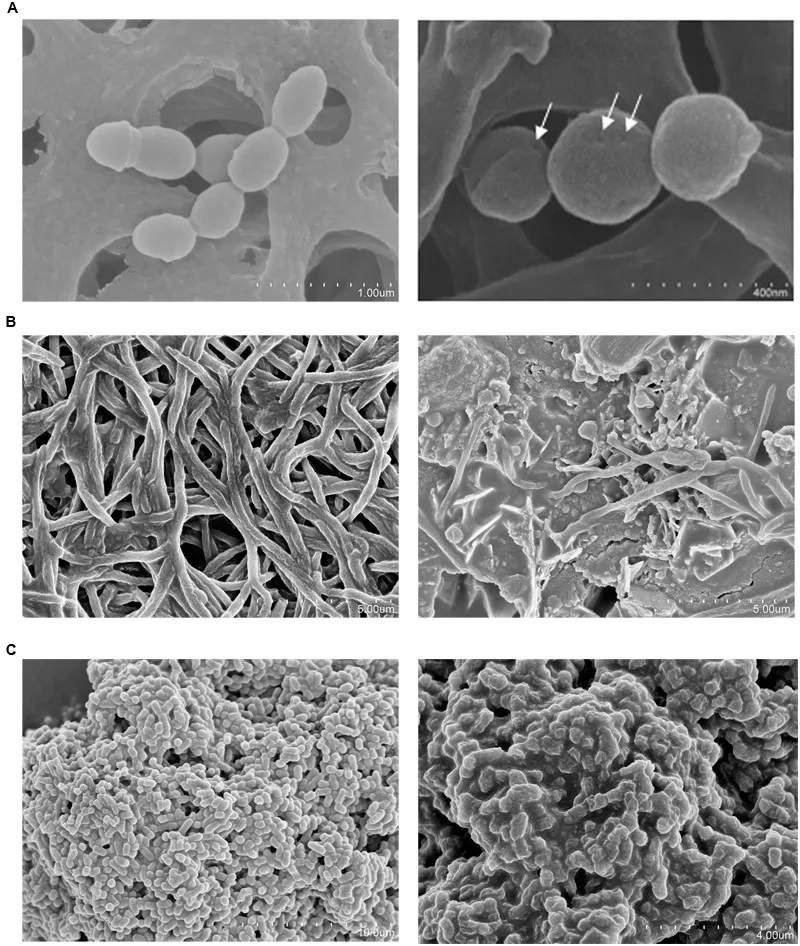
The first is the production of bacteriocins — antimicrobial peptides that inhibit the growth of S. mutans, S. sobrinus, Prevotella intermedia and other oral pathogens. Llena et al. (2019) demonstrated that S. dentisani supernatant inhibits 20 of the 22 microbial species tested, including those implicated in endodontic infections [4]. Electron microscopy images reveal structural changes in bacterial membranes consistent with bacteriocin activity.
Bacteriocins are ribosome-synthesised — a mechanism of action reminiscent of lariocidin. They are active at low concentrations and often stable at extreme temperatures and pH. Many act by depolarising the target bacterium’s cytoplasmic membrane, causing cell death through pore formation.
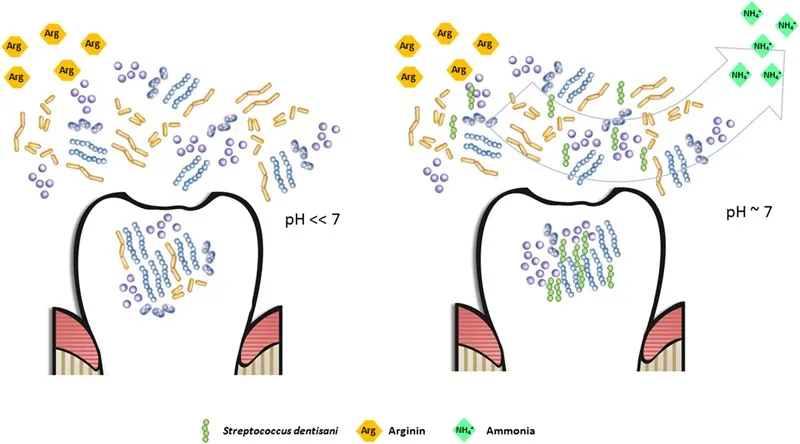
The second mechanism is an arginolytic pathway: S. dentisani metabolises arginine to produce ammonium, which buffers the acidic pH of dental plaque. This is decisive, because caries is not caused directly by bacteria, but by the acid they produce. Neutralising the acid means breaking the causal chain at its root.
Global distribution and correlation with oral health
S. dentisani is not a local exception. López-Santacruz et al. (2021) analysed 16S rRNA datasets from multiple continents, demonstrating that the bacterium is present in the saliva and plaque of geographically distant populations [5]. Typical salivary levels range from 10³ to 10⁵ cells/ml.
The most relevant finding: in a cohort of Mexican children, S. dentisani proportions in supragingival plaque were significantly higher in caries-free subjects than in those with active caries (p = 0.002), with a negative correlation between dmft index and bacterial percentage (p < 0.001).
Similar results emerged from a study on Colombian children, where S. dentisani was identified in all samples examined, with significant differences related to the use of fluoride products and food intake frequency [6].
Biofilm and competition strategies
Bedoya-Correa et al. (2024) tested the probiotic capacity of S. dentisani against biofilms of S. mutans, S. mitis and Candida albicans using three strategies: competition, exclusion and displacement [7]. The clearest result came from the displacement strategy — dislodging already-formed pathological biofilms — where S. dentisani significantly reduced streptococcal biofilms. Against C. albicans, the effect was evident in both competition and displacement.
Conrads et al. (2019) added a layer of complexity: S. dentisani strains with similar bacteriocin gene clusters can coexist thanks to cross-immunity genes, while dissimilar strains inhibit each other [8]. This suggests a refined ecological dynamic, where intraspecific diversity regulates colonisation — a principle also seen in the evolution of periodontal microbial complexes.
The other side: fibrils and endocarditis
No bacterium is purely good. Ronis et al. (2019) showed that S. oralis subsp. dentisani produces monolateral fibrils composed of serine-rich repeat proteins (SRRPs) — named FapA, FapB and FapC — that mediate saliva adhesion through sialic acid binding [9]. The same structures were found in clinical isolates from patients with infective endocarditis.
This is the paradox of oral commensals in the mitis group: in plaque they are model citizens, but if they reach the bloodstream — through an extraction, aggressive probing, even brushing in the presence of gingival bleeding — they can colonise heart valves. The fibrils that anchor them to saliva also anchor them to platelets. It is the same fine line between infection and chronic disease we observe in periodontology.
This does not invalidate the probiotic potential of S. dentisani, but it demands caution: an effective oral probiotic must remain confined to the oral cavity.
The honest limitation
Not everything is resolved. Conrads’ group observed the development of resistance and adaptation to S. dentisani bacteriocins during in vitro experiments [8]. An important warning: pathogens learn. Probiotic strains will need continuous monitoring to ensure their efficacy over time.
Moreover, most of the evidence is in vitro or observational. Randomised clinical trials are needed to define dosages, delivery vehicles and long-term impact on the oral microbiome as a whole.
From Valencia to the clinic
Strain 7746 has been patented under the name AB-Dentisanium® and represents the most advanced candidate for a site-specific oral probiotic against caries. Unlike gut probiotics repurposed for the oral cavity, S. dentisani comes from the mouth and returns to the mouth — with an evolutionary advantage in colonising the target site. López-López et al. demonstrated that the bacterium does not survive simulated gastric digestion, limiting the risk of systemic effects [3].
The idea is simple and powerful: rather than killing pathogenic bacteria with antimicrobials that devastate the entire ecosystem, strengthen the population of the good ones. A paradigm shift — from destruction to ecology.
References
-
Camelo-Castillo A, Benítez-Páez A, Belda-Ferre P, et al. Streptococcus dentisani sp. nov., a novel member of the mitis group. Int J Syst Evol Microbiol. 2013;64(Pt 1):60-65. DOI
-
Jensen A, Scholz CFP, Kilian M. Re-evaluation of the taxonomy of the Mitis group of the genus Streptococcus […]. Int J Syst Evol Microbiol. 2016;66(11):4803-4820. DOI
-
López-López A, Camelo-Castillo A, Ferrer MD, Simon-Soro Á, Mira A. Health-Associated Niche Inhabitants as Oral Probiotics: The Case of Streptococcus dentisani. Front Microbiol. 2017;8:379. DOI
-
Llena C, Almarche A, Mira A, López MA. Antimicrobial efficacy of the supernatant of Streptococcus dentisani against microorganisms implicated in root canal infections. J Oral Sci. 2019;61(1):184-194. DOI
-
López-Santacruz HD, López-López A, Revilla-Guarinos A, et al. Streptococcus dentisani is a common inhabitant of the oral microbiota worldwide and is found at higher levels in caries-free individuals. Int Microbiol. 2021;24(4):619-629. DOI
-
Angarita-Díaz MP, Díaz JA, Tupaz HA, et al. Presence of Streptococcus dentisani in the dental plaque of children from different Colombian cities. Clin Exp Dent Res. 2019;5(3):184-190. DOI
-
Bedoya-Correa CM, Betancur-Giraldo S, Franco J, Arango-Santander S. Probiotic Effect of S. dentisani on Oral Pathogens: An In Vitro Study. Pathogens. 2024;13(5):351. DOI
-
Conrads G, Westenberger J, Lürkens M, Abdelbary MMH. Isolation and Bacteriocin-Related Typing of S. dentisani. Front Cell Infect Microbiol. 2019;9:110. DOI
-
Ronis A, Brockman K, Singh AK, et al. Streptococcus oralis subsp. dentisani Produces Monolateral Serine-Rich Repeat Protein Fibrils, One of Which Contributes to Saliva Binding via Sialic Acid. Infect Immun. 2019;87(10):e00406-19. DOI
FAQ
- What is Streptococcus dentisani?
- Streptococcus dentisani is a bacterial species isolated from the dental plaque of caries-free individuals. It belongs to the mitis group of streptococci and was formally described in 2013 by Alex Mira's group at FISABIO (Valencia, Spain). It produces bacteriocins against major oral pathogens and buffers acidic pH through arginine metabolism.
- How does the probiotic action of S. dentisani work?
- S. dentisani acts through a dual mechanism: it produces bacteriocins that inhibit the growth of Streptococcus mutans and other cariogenic pathogens, and it possesses an arginolytic pathway that generates ammonium from arginine, buffering plaque acidity — the primary cause of enamel demineralisation.
- Is Streptococcus dentisani found in all people?
- Studies across multiple continents have detected S. dentisani in the saliva and supragingival plaque of the majority of individuals examined, with significantly higher levels in caries-free subjects compared to those with active carious lesions.
- Can Streptococcus dentisani be dangerous?
- Like other mitis-group streptococci, S. dentisani has surface fibrils (serine-rich repeat proteins) that can mediate platelet adhesion. Clinical isolates of S. oralis subsp. dentisani have been found in patients with infective endocarditis. The risk is linked to transient bacteraemia — bacteria entering the bloodstream — not to their presence in dental plaque.
- Are there products containing Streptococcus dentisani?
- Strain S. dentisani 7746 has been patented under the trade name AB-Dentisanium® and is the subject of clinical studies as an oral probiotic. Further research is needed to define dosages, delivery vehicles and long-term impact on the oral microbiome.
Looking for a specialist?
Parodontologia a Frosinone →Prevenzione e cura delle malattie gengivali e parodontali
Need a professional opinion?
Book an appointment at Dr. Bruschi's practice in Frosinone. First visit includes full diagnosis and personalised treatment plan.
Stay Updated
New articles on periodontology, implantology and oral surgery — delivered to your inbox.
Comments
Loading comments...
Leave a comment