The dental X-ray that could save your bones: how AI turns the OPT into an osteoporosis screening tool
Leggi in Italiano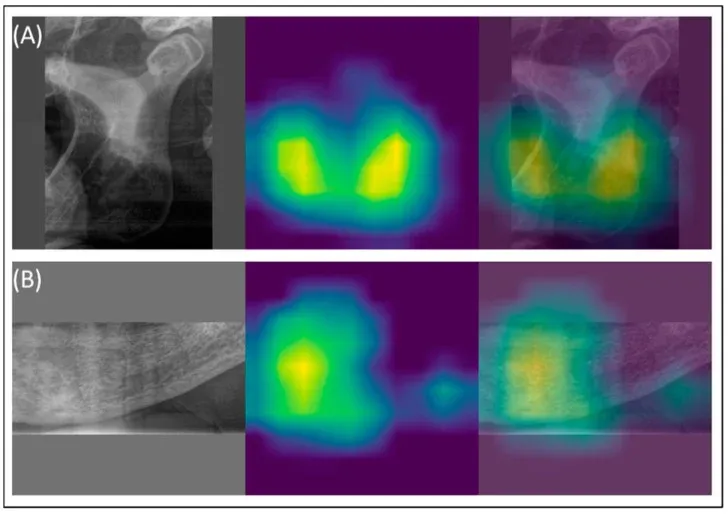
In brief — Two meta-analyses (2024–2025) demonstrate that AI can detect osteoporosis on panoramic dental X-rays with pooled sensitivity of 88%, versus 35% for manual assessment. The dental office — already the most frequently visited healthcare touchpoint — could become a first-line bone screening point at near-zero extra cost.
Sintesi (IT) — Due meta-analisi (2024–2025) dimostrano che l’AI può rilevare l’osteoporosi sull’ortopantomografia con sensibilità pooled dell’88%, contro il 35% della valutazione manuale. Lo studio dentistico potrebbe diventare una prima linea di screening osseo senza costi aggiuntivi per il paziente.
The 500-million paradox
Five hundred million people worldwide live with osteoporosis. Most don’t know it. They have no diagnosis.
Not because the tests don’t exist. Not because doctors aren’t skilled. But because osteoporosis is an insidious and silent disease: it doesn’t burn, doesn’t swell, doesn’t cause fever. It only makes itself known when something breaks — a vertebra collapsing under the weight of a shopping bag, a femur fracturing in a trivial fall. By that point the damage is already advanced, and treatment works uphill.
Every three seconds in the world, a fragility fracture related to osteoporosis occurs. In Europe, one in three women and one in five men over fifty will suffer at least one osteoporotic fracture in their lifetime. Yet DXA — the gold-standard radiological exam for measuring bone mineral density — is too often prescribed only after the first fracture, when it would have been far better to act years earlier.
The question arises naturally: is there a simple way to detect this disease before it makes itself felt?
The answer, emerging from two meta-analyses published between 2024 and 2025, comes from an unexpected place: your dentist’s office.
That X-ray you get as routine
The orthopantomogram — OPT or OPG, commonly called a “panoramic X-ray” — is the radiograph taken routinely in dental offices every one or two years. In a single image it shows all the teeth, the maxillary and mandibular bones, the paranasal sinuses, and the temporomandibular joints. It is one of the most common exams in medicine: millions of OPTs are performed every year, at a cost of roughly €30–70.
What most patients don’t know is that in that image there is far more than teeth.
The inferior border of the mandible — the so-called cortical plate or mandibular inferior cortex — reflects the bone health of the entire skeleton. Mandibular bone responds to the same hormonal, nutritional, and metabolic variations that affect the spine and femur. When systemic bone density decreases, so does the thickness and regularity of the mandibular cortex.
Radiologists have known this for decades. As early as the 1990s, specific morphometric indices were developed — the Mandibular Cortical Index (MCI), the Mandibular Cortical Width (MCW), the Panoramic Mandibular Index (PMI) — to visually assess these parameters on OPT and estimate osteoporosis risk.
The problem was precision.
Scientific box — The limits of the human eye
A 2019 meta-analysis published in Dentomaxillofacial Radiology (Kinalski et al., PMID 31596133, 33 studies, 5,266 women) measured the performance of the Mandibular Cortical Index assessed manually by experienced radiologists:
- Sensitivity for osteoporosis: 35%
- Specificity: 88%
Translated: out of 100 women with osteoporosis, the human eye correctly identified only 35. The other 65 were missed.
High specificity means few false positives — but 35% sensitivity is far too low for a screening tool.
Artificial intelligence enters the picture
Over the past six years, dozens of research groups worldwide have trained deep learning algorithms — convolutional neural networks (CNNs) in variants such as ResNet, EfficientNet, VGG-16, and GoogLeNet — to analyze OPTs and automatically classify osteoporosis risk, comparing results against DXA as the gold standard.
The results surprised even the researchers.
In 2019, a Korean group (Lee et al., Dentomaxillofacial Radiology, PMID 30004241, 1,268 patients) published a study using a multi-column DCNN that achieved an AUC of 0.9987 — near-perfect diagnostic accuracy under controlled conditions. An extraordinary result, but on a single dataset.
Then came the meta-analyses, which aggregate data from dozens of studies and produce more robust, generalizable estimates.
Scientific box — The two key meta-analyses
Ghasemi et al. (2025) — Journal of Dentistry, 24 studies included
- Pooled sensitivity: 87.9%
- Pooled specificity: 81.9%
- Diagnostic Odds Ratio: 32.99
Khadivi et al. (2024) — Osteoporosis International, 22 studies included
- DCNN: Sensitivity 87%, Specificity 92%
- Conventional algorithms: Sens. 82%, Spec. 96%
Both meta-analyses compare AI results against DXA as the gold standard.
From 35% with the human eye to 88% with AI: more than double the diagnostic effectiveness, on the same exam, at no extra cost to the patient.
The 92% specificity is particularly relevant in a screening context: it means the system generates few false positives, avoiding unnecessary alerts in healthy patients and preventing the healthcare system from being overloaded with unneeded DXA referrals.
How AI “sees” — and where it looks
One of the most fascinating aspects of this research concerns how the models actually work. Thanks to visualization techniques called GradCAM (Gradient-weighted Class Activation Mapping), it is possible to see literally which zones of the image attract the neural network’s attention when classifying a case as osteoporotic.
The answer is precise: the inferior border of the mandible and the mental foramen (the opening through which the mental nerve passes). Exactly the same anatomical regions an experienced radiologist would examine.
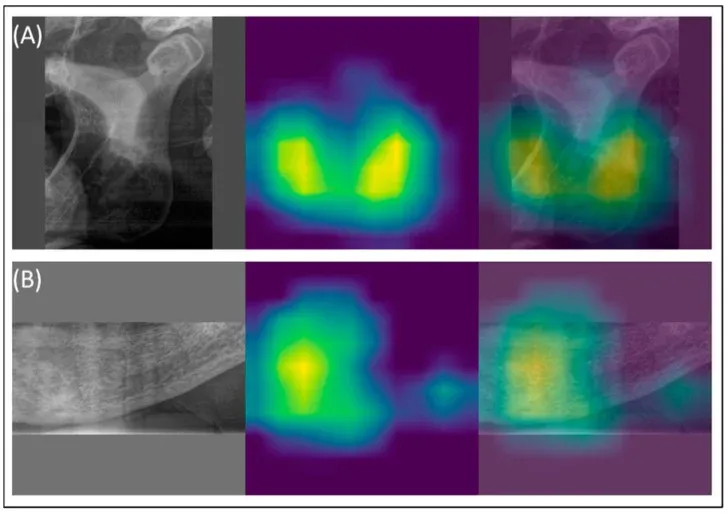
AI did not learn an arbitrary statistical shortcut. It learned the same biology clinicians have known for decades — only with a speed and consistency the human eye cannot match.
A study from the Charité group in Berlin (Gaudin et al., 2024, Diagnostics, PMID 39451621) proposed a hybrid approach: YOLOv8 to automatically identify regions of interest in the image, then EfficientNet for classification. Result: sensitivity 83%, AUC 0.76. Not the highest value in absolute terms, but achieved with explicit methodological rigor — including external validation — that many previous studies lacked.
Did you know — The Italian data
In 2025, an Italian team published in Journal of Clinical Medicine (Fanelli et al., PMID 40648835, universities of Foggia and Parma, with external validation) the first study with external validation on an Italian dataset.
The ResNet-50 model, trained on 245 OPTs from postmenopausal women and externally validated on 56 cases from Parma, achieved:
- Sensitivity: 90.5%
- AUC: 0.786
High sensitivity means few osteoporosis cases missed — the most important quality for a first-line screening tool.
The dentist as systemic sentinel
There is an epidemiological figure that rarely enters the clinical debate, but that completely changes the perspective.
In the United States, according to the American Dental Association, 65% of adults visit the dentist at least once a year. Only 43% visit a GP with the same frequency. European data (Eurobarometer 2022) show an average of 1.5 dental visits per year per EU citizen.
The dental office is already, de facto, the most capillary access point to the healthcare system — more so than the general practitioner. Every year millions of people walk into that office, undergo a routine OPT, and leave with clinical information that stops at the teeth. Yet the dental office is already a place where systemic disease signals are detected — from periodontal bacteria reaching the brain to osteoporosis markers in the mandibular cortex.
Integrating an AI bone-analysis module into an already-performed OPT has near-zero marginal cost for the patient. It adds no radiation (the OPT is already part of the dental pathway), requires no second appointment, imposes no invasive procedure. It requires an algorithm and a workflow.
In cost-effectiveness terms: DXA costs three to eight times more than an OPT. And it requires dedicated equipment, trained staff, and a medical prescription that millions of at-risk people never receive.
For professionals — Index comparison
Structured comparison of OPT assessment methods for osteoporosis screening:
Method Sensitivity Specificity Notes Manual MCI 35% 88% Kinalski 2019 meta-analysis, 5,266 women MCW ≤3 mm 42–60% 82–90% Good specificity, low sensitivity Manual MCI + FRAX >15% High ~95% Optimal pre-AI combination AI on OPT (pooled) 87–88% 82–95% 2024–2025 meta-analyses MCI + alveolar density (AI) — — External validation AUC 0.97–1.00 (Okubo 2025) The AI advantage is quantifiable: this is not a marginal improvement — it is a categorical leap in diagnostic sensitivity.
The current limitation — and why it matters to say so
The research is solid. But intellectual honesty demands saying where we actually stand.
The most recent systematic review on the topic — Querrer et al., published in Dentomaxillofacial Radiology in 2026 (PMID 40580938), 13 studies analyzed — identified a critical gap: not one of the analyzed studies integrated these models into a real dental office setting. All results come from retrospective datasets, under controlled experimental conditions.
The Charité group raised another issue: many of the most optimistic results in the literature (AUC >0.99) reflect methodological biases — unbalanced patient selection, lack of external validation, comparison between groups that are too far apart (young healthy patients vs. elderly osteoporotic ones). When more rigorous methodology is applied, performance drops — remaining clinically relevant, but far from “perfection.”
The technology is ready. The scientific rigor is being consolidated. What is missing is the third element: the bridge between laboratory and dental office. Accessible interfaces, workflows integrated into practice management software, specialist referral protocols, clinician training.
This is the frontier the field is working on — and where, in the coming years, we will see the first real clinical results.
Conclusion
The orthopantomogram is one of the most common medical exams in the world. For decades it did one job: show us teeth. Today we know that image contains much more.
Artificial intelligence has transformed a manual assessment with 35% sensitivity into a screening tool with pooled sensitivity of 88% — aggregated across dozens of studies and tens of thousands of patients. This is not science fiction: it is literature published in Osteoporosis International, Journal of Dentistry, and Scientific Reports.
The next step is not technological. It is organizational: integrating this tool into daily clinical practice, training dentists to recognize skeletal warning signs, building referral protocols between the dental office and the osteoporosis specialist.
When that step is taken, the dental visit will no longer be just a tooth check. It will also be — finally — a front line for bone health.
Frequently Asked Questions
Can the OPT replace DXA?
No. The AI-analyzed OPT is a first-level screening tool — it identifies at-risk patients who should then be referred for DXA to obtain a definitive diagnosis. It does not replace it.
Can my dentist already do this?
In routine Italian clinical practice, no. AI models for bone analysis of OPT are not yet integrated into commercial dental software. Some university centers may have them in experimental use.
What can I do right now?
If you are over 50, postmenopausal, have had a fracture, or have family members with osteoporosis, speak with your GP about the possibility of having a DXA scan. Don’t wait for an algorithm to tell you there’s a problem.
Why does the mandible reflect systemic bone health?
Mandibular bone responds to the same hormonal, metabolic, and nutritional changes that affect the spine and femur. Estrogen loss at menopause, vitamin D deficiency, osteoclast hyperactivity — all these mechanisms act on the entire skeleton, mandible included.
References
- Ghasemi et al. (2025) — Journal of Dentistry — PMID 40010536
- Khadivi et al. (2024) — Osteoporosis International — PMID 39177815
- Fanelli et al. (2025) — Journal of Clinical Medicine — PMID 40648835
- Querrer et al. (2026) — Dentomaxillofacial Radiology — PMID 40580938
- Kinalski et al. (2019) — Dentomaxillofacial Radiology — PMID 31596133
- Gaudin et al. (2024) — Diagnostics — PMID 39451621
- Turosz et al. (2023) — Dentomaxillofacial Radiology — PMID 37665008
- Lee et al. (2019) — Dentomaxillofacial Radiology — PMID 30004241
FAQ
- Can the OPT replace DXA?
- No. The AI-analyzed OPT is a first-level screening tool — it identifies at-risk patients who should then be referred for DXA to obtain a definitive diagnosis. It does not replace it.
- Can my dentist already do this?
- In routine Italian clinical practice, no. AI models for bone analysis of OPT are not yet integrated into commercial dental software. Some university centers may have them in experimental use.
- What can I do right now?
- If you are over 50, postmenopausal, have had a fracture, or have family members with osteoporosis, speak with your GP about the possibility of having a DXA scan.
- Why does the mandible reflect systemic bone health?
- Mandibular bone responds to the same hormonal, metabolic, and nutritional changes that affect the spine and femur. Estrogen loss, vitamin D deficiency, and osteoclast hyperactivity all act on the entire skeleton — the mandible included.
References
- https://pubmed.ncbi.nlm.nih.gov/40010536/
- https://pubmed.ncbi.nlm.nih.gov/39177815/
- https://pubmed.ncbi.nlm.nih.gov/40648835/
- https://pubmed.ncbi.nlm.nih.gov/40580938/
- https://pubmed.ncbi.nlm.nih.gov/31596133/
- https://pubmed.ncbi.nlm.nih.gov/39451621/
- https://pubmed.ncbi.nlm.nih.gov/37665008/
- https://pubmed.ncbi.nlm.nih.gov/30004241/
Looking for a specialist?
Innesto Osseo a Frosinone →Tipi di innesto, tecniche e indicazioni per ricostruire l'osso perduto
Need a professional opinion?
Book an appointment at Dr. Bruschi's practice in Frosinone. First visit includes full diagnosis and personalised treatment plan.
Stay Updated
New articles on periodontology, implantology and oral surgery — delivered to your inbox.
Comments
Loading comments...
Leave a comment