The menopause-gum axis that medicine is rediscovering
Leggi in Italiano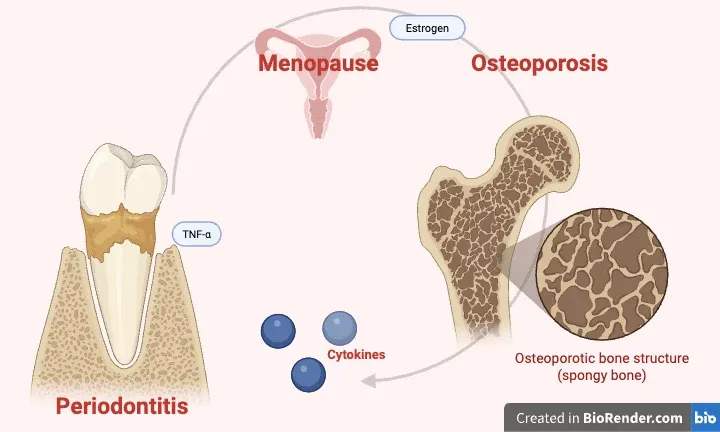
In brief — A 2023 meta-analysis on 19,611 patients shows postmenopausal women with osteoporosis have more severe periodontitis (+0.89 mm clinical attachment loss) than those without. A Korean study on 2,573 women identifies the first four years after menopause as the critical window: periodontal disease risk doubles. The two conditions share the same inflammatory biology.
Sintesi (IT) — Una meta-analisi 2023 su 19.611 pazienti dimostra che le donne in post-menopausa con osteoporosi hanno parodontite più grave (+0,89 mm di perdita di attacco clinico) rispetto a quelle senza. Uno studio coreano su 2.573 donne identifica nei primi 4 anni dopo la menopausa la finestra critica: il rischio di malattia parodontale raddoppia. Le due condizioni condividono la stessa biologia infiammatoria.
Two diseases, one biology
Think of someone you know who learned they had osteoporosis — maybe after a bad fracture or a vertebral collapse.
Now think of someone with gum problems. In your mind, they’re probably two different people, on two different care paths.
And yet they could be the same person.
The correlation between osteoporosis and periodontal disease is one of the most fertile — and most underappreciated — chapters of contemporary medicine. Fertile because the data are accumulating fast, with patient numbers now in the tens of thousands. Underappreciated because the historic boundary between medicine and dentistry slows the diffusion of these findings to the people who need them most: the patients themselves.
This article is an attempt to dismantle that boundary, as has already been done with others — backed by data.
The number that changes the perspective
In 2023, a meta-analysis published in Heliyon (Qi et al., PMID 37920517) aggregated data from 28 observational studies, totaling 19,611 patients. The aim: to answer a precise question — do postmenopausal women with osteoporosis have similar periodontal conditions, or worse, compared to those without?
The answer may surprise you: periodontitis is more severe in the osteoporosis group — and not by a little.
Compared to women without osteoporosis, those with the diagnosis showed:
- +0.89 mm clinical attachment loss (CAL) — the direct measure of damage to the periodontal tissue
- +0.27 mm probing depth (PD) — an index of active inflammation
- +0.28 mm gingival recession (GR)
- +12.71% bleeding on probing (BOP) — the classical sign of gum inflammation
- significantly lower bone mineral density (BMD) also at dental sites
Singly, any of these numbers might look like an abstract numerical difference. But 0.89 mm of additional clinical attachment loss, across nearly twenty thousand patients, represents a clinically meaningful gap: in many patients, it could be the difference between mild and moderate periodontitis, between a tooth that is saved and one with hopeless prognosis.
Scientific box — What we measure when we measure periodontitis
Clinical attachment loss (CAL) is the principal measure of periodontal disease progression. It indicates how many millimeters the tissue still “holding” the tooth has detached from the root surface. CAL ≥3 mm is the clinical threshold for periodontitis; ≥5 mm indicates moderate-to-severe disease.
Bleeding on probing (BOP) is the gum tissue’s response to the diagnostic probe: when it bleeds, inflammation is active. BOP >25% is associated with progression of periodontal damage.
Probing depth (PD) measures the depth of the gingival pocket: beyond 4 mm, home oral hygiene becomes ineffective and inflammation tends to chronicize.
The critical window: the four years after menopause
If the meta-analysis gives the size of the phenomenon, a Korean study published in PLoS ONE (Lee Y., 2022, PMID 35298563) illuminates its temporal dynamics — and the result is perhaps even more clinically relevant.
The study analyzed Korea National Health and Nutrition Examination Survey (KNHANES) data on 2,573 menopausal women, stratifying the sample by years since the last menstrual period.
Overall, the risk of periodontal disease in osteoporotic women was already significantly elevated: OR = 1.25 (95% CI: 1.00–1.56). But when women within the first four years after menopause were analyzed separately, the risk nearly doubled:
OR* = 2.08 (95% CI: 1.15–3.77)
*The Odds Ratio (OR) is a statistical measure that quantifies the strength of association between two events.
Translated clinically: a woman with osteoporosis less than four years past menopause has roughly twice the odds of developing periodontal disease compared to a non-osteoporotic woman at the same stage.
This finding has immediate practical implications: the menopausal transition is the temporal window in which the interaction between the two diseases is most intense — and therefore the moment in which prevention and early diagnosis have the highest potential payoff.
Did you know — The gender factor
A Taiwanese epidemiological study based on the national database (Lin et al., 2015, Medicine, PMID 25700325) analyzed the periodontitis–osteoporosis relationship separately for men and women.
Result: in women, the risk was significantly elevated — OR 1.96 (95% CI: 1.17–3.26). In men, the association was not statistically significant.
This does not mean men are immune. It means that in women the hormonal system — and specifically the estrogen drop — is a powerful amplifier of the shared risk between the two diseases.
Why it happens: shared biology
The correlation between osteoporosis and periodontitis is not statistical coincidence. It has a precise biological explanation, progressively clarified by recent research.
Both diseases originate from an imbalance in bone remodeling: the balance between osteoblasts (cells that build bone) and osteoclasts (cells that break it down) tilts toward demolition. In osteoporosis, this imbalance is systemic — it affects the entire skeleton. In periodontitis, it is localized to the alveolar bone, the bone that holds the teeth. The causes differ, but the final molecular mechanisms are similar.
The pivot is chronic inflammation.
Scientific box — The cytokines that do the damage
Three inflammatory molecules play a central role in both conditions:
- IL-1β (Interleukin-1β): a potent stimulator of osteoclast activity; elevated both in the gingival crevicular fluid of periodontitis patients and in the serum of osteoporosis patients.
- TNF-α (Tumor Necrosis Factor alpha): promotes bone resorption and inhibits osteoblastic formation; in both diseases, serum levels are chronically elevated.
- IL-6 (Interleukin-6): a key regulator of the RANK/RANKL/OPG axis governing bone turnover; its overproduction accelerates resorption both alveolar and systemic.
Source: Yu B, Wang CY, Periodontology 2000, 2022, PMID 35244945.
Periodontitis is not just a local mouth disease. The bacteria of the subgingival biofilm and the inflammatory mediators they produce enter systemic circulation and reach distant organs — and when they do, they raise the levels of these same markers in blood. In an organism already in a chronic inflammatory state — like a postmenopausal woman with osteoporosis — this contribution adds to the existing one, worsening both conditions.
This is the meaning of the bidirectional relationship: osteoporosis increases susceptibility to alveolar bone loss, and untreated periodontitis fuels the systemic inflammation that in turn accelerates skeletal resorption. The two diseases feed each other.
A 2026 review by Maria Contaldo (University of Campania Luigi Vanvitelli, in Advances in Experimental Medicine and Biology — PMID 41225098) identifies microbiota dysbiosis — both oral and gut — as a critical element in both conditions: perturbation of the shared microbial ecosystem creates the conditions for maintaining a chronic inflammatory state that resolves neither at the gum level nor at the systemic skeletal level.
The role of estrogens
The estrogen drop at menopause does not act only on bones. It acts on gums too.
Estrogens modulate the local immune response in the oral cavity, microbiota composition, alveolar bone density, and the activity of collagenase — the enzyme that degrades the collagen of tooth-supporting tissues. When estrogen levels fall sharply, as happens in the first years after menopause, these protective effects diminish simultaneously across all fronts.
A 2025 review in Cureus (Jawed et al., PMID 40462880) systematically analyzed the link between female hormonal variations and gum health, confirming that the menopausal transition represents a phase of increased periodontal vulnerability — independent of oral hygiene and pre-existing dental status.
This finding has an often-overlooked practical implication: a woman entering menopause should discuss with her dentist not only bone density, but also gum health — and vice versa. The two prevention paths should not be parallel and separate, but integrated.
Did you know — Shared modifiable risk factors
Osteoporosis and periodontitis share more than biology. They share modifiable risk factors:
- Vitamin D deficiency: lowers systemic bone mineralization and increases susceptibility to periodontal inflammation.
- Cigarette smoking: accelerates bone resorption and impairs gum vascularization, masking bleeding (false reassurance).
- Sedentary lifestyle: reduces the mechanical stimulus on bone needed to maintain bone mass.
- Diet poor in calcium and protein: depletes both systemic and alveolar bone structure.
Acting on these factors lowers the risk of both diseases at once.
What changes in practice
Research on this correlation has not yet fully entered daily clinical practice — neither medical nor dental. Yet the available data already justify concrete changes in how we manage these patients.
For the dentist. A menopausal patient with a diagnosis of osteoporosis is, by definition, a high-risk periodontal patient. She deserves more frequent monitoring, a lower alert threshold, and active coordination with the GP or rheumatologist managing her osteoporosis. Periodontal parameters — CAL, BOP, PD — are not just dental data: in this context they become indicators of systemic risk. This is also where AI analysis of dental panoramic X-rays can add a layer of bone screening to the routine dental visit.
For the physician. A patient with osteoporosis who has not had a periodontal checkup in the last twelve months has a gap in their prevention pathway. Referral to the dentist — not just for caries control but specifically for periodontal evaluation — should be part of the standard treatment plan.
For the patient. If you are menopausal and have a diagnosis of osteoporosis, or even just reduced bone density, know that your gums are in a phase of increased vulnerability. Don’t wait for pain or bleeding to see your dentist: by that point inflammation has already been active for some time.
Conclusion
Fifty years ago, the idea that gums and the spine or femoral head could “speak” the same biological language would have sounded speculative. Today we have 19,611 patients from a meta-analysis confirming it, studies on tens of thousands of women identifying the critical temporal window, and a molecular understanding sufficiently detailed to explain the why.
The challenge now is not scientific. It is organizational and cultural: getting used to looking at the mouth as part of the body, and the body as a single system in which gums, bones, hormones, and inflammation influence each other reciprocally.
Next time you visit your dentist — or refer a patient — keep it in mind.
Frequently Asked Questions
If I have osteoporosis, will I definitely develop periodontitis?
No. Statistical association is not clinical determinism. Having osteoporosis raises your risk, but it does not guarantee periodontitis will develop — especially with good oral hygiene and regular checkups.
If I treat periodontitis, will my osteoporosis improve too?
Evidence on this front is still preliminary. Some studies suggest that treating periodontitis lowers systemic inflammatory markers — including those tied to bone resorption — but no randomized clinical trials have yet shown a direct improvement in bone mineral density from periodontal therapy alone.
Does the relationship apply to men too?
The strongest evidence concerns postmenopausal women, due to the amplifying role of estrogens. In men the association exists but is less studied and statistically less robust. That doesn’t mean it isn’t relevant — it means we still lack adequately powered male-cohort studies.
Should I worry if my gums just bleed?
Gum bleeding is always a sign of active inflammation. It does not automatically mean severe periodontitis, but it is always a valid reason for a dental evaluation. In a context of menopause and reduced bone density, it deserves even closer attention.
References
- Qi J et al. (2023) — Heliyon — PMID 37920517 — meta-analysis, 28 studies, 19,611 patients
- Lee Y. (2022) — PLoS ONE — PMID 35298563 — KNHANES, 2,573 women
- Yu B, Wang CY (2022) — Periodontology 2000 — PMID 35244945 — shared inflammatory mechanisms
- Contaldo M. (2026) — Adv Exp Med Biol — PMID 41225098 — bidirectional relationship and dysbiosis
- Lin TH et al. (2015) — Medicine — PMID 25700325 — Taiwanese cohort, gender factor
- Jawed STM et al. (2025) — Cureus — PMID 40462880 — hormonal variations and gum health
FAQ
- If I have osteoporosis, will I definitely develop periodontitis?
- No. Statistical association is not clinical determinism. Having osteoporosis raises your risk, but it does not guarantee periodontitis will develop — especially with good oral hygiene and regular checkups.
- If I treat periodontitis, will my osteoporosis improve too?
- Evidence on this front is still preliminary. Some studies suggest that treating periodontitis lowers systemic inflammatory markers — including those tied to bone resorption — but no randomized clinical trials have yet shown a direct improvement in bone mineral density from periodontal therapy alone.
- Does the relationship apply to men too?
- The strongest evidence concerns postmenopausal women, due to the amplifying role of estrogens. In men the association exists but is less studied and statistically less robust. That doesn't mean it isn't relevant — it means we still lack adequately powered male-cohort studies.
- Should I worry if my gums just bleed?
- Gum bleeding is always a sign of active inflammation. It does not automatically mean severe periodontitis, but it is always a valid reason for a dental evaluation. In a context of menopause and reduced bone density, it deserves even closer attention.
References
Looking for a specialist?
Parodontologia a Frosinone →Diagnosi e trattamento della parodontite nello Studio Denti Più
Need a professional opinion?
Book an appointment at Dr. Bruschi's practice in Frosinone. First visit includes full diagnosis and personalised treatment plan.
Stay Updated
New articles on periodontology, implantology and oral surgery — delivered to your inbox.
Comments
Loading comments...
Leave a comment